Scalable neutral H2O2 electrosynthesis by platinum diphosphide nanocrystals by regulating oxygen reduction reaction pathways | Nature Communications

of H2O2 production activity and stability a, Kinetic current density.... | Download Scientific Diagram

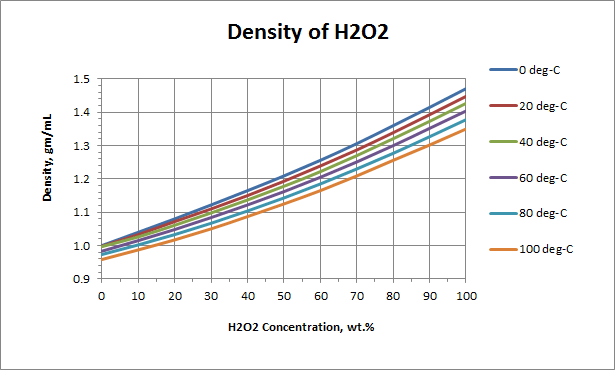
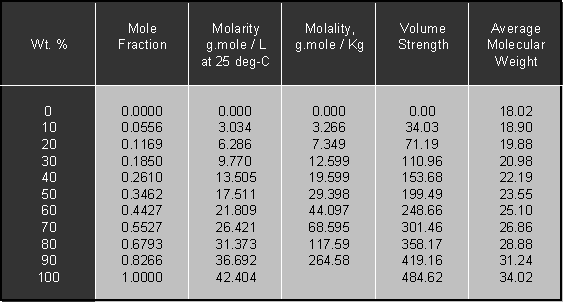
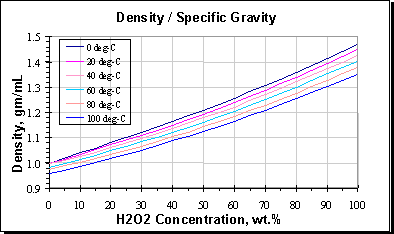
Density or Specific Gravity of Hydrogen Peroxide; Ref: M.F. Easton,... | Download Scientific Diagram

OneClass: If I have hydrogen peroxide, molecular weight(g/mol) is 34.02, its density (g/ml) is 1.2, i...
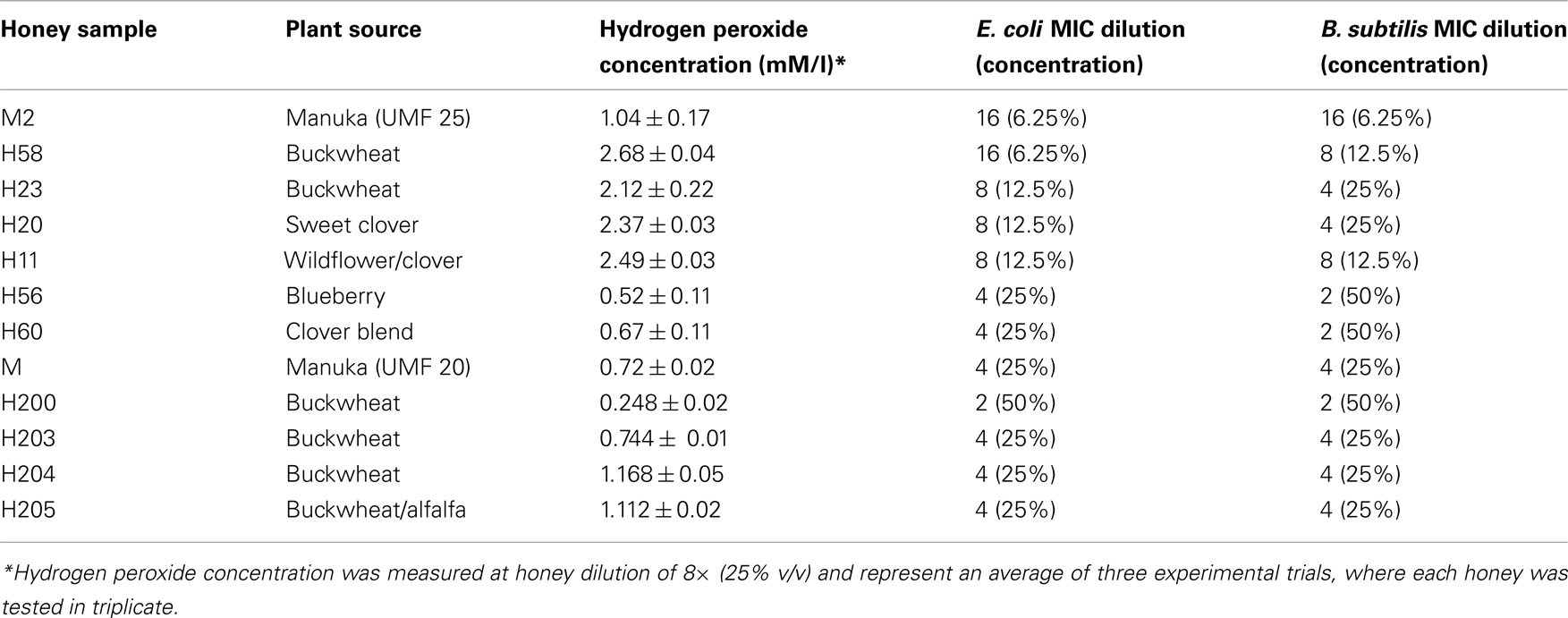
Frontiers | Re-Examining the Role of Hydrogen Peroxide in Bacteriostatic and Bactericidal Activities of Honey

Concentration of Hydrogen Peroxide in a 10 Volume Solution - An Interesting Stoichiometry Problem - YouTube
Bottles of Hydrogen Peroxide say '3%', but what exactly does that mean? What is the molarity of 3% H2O2? - Quora
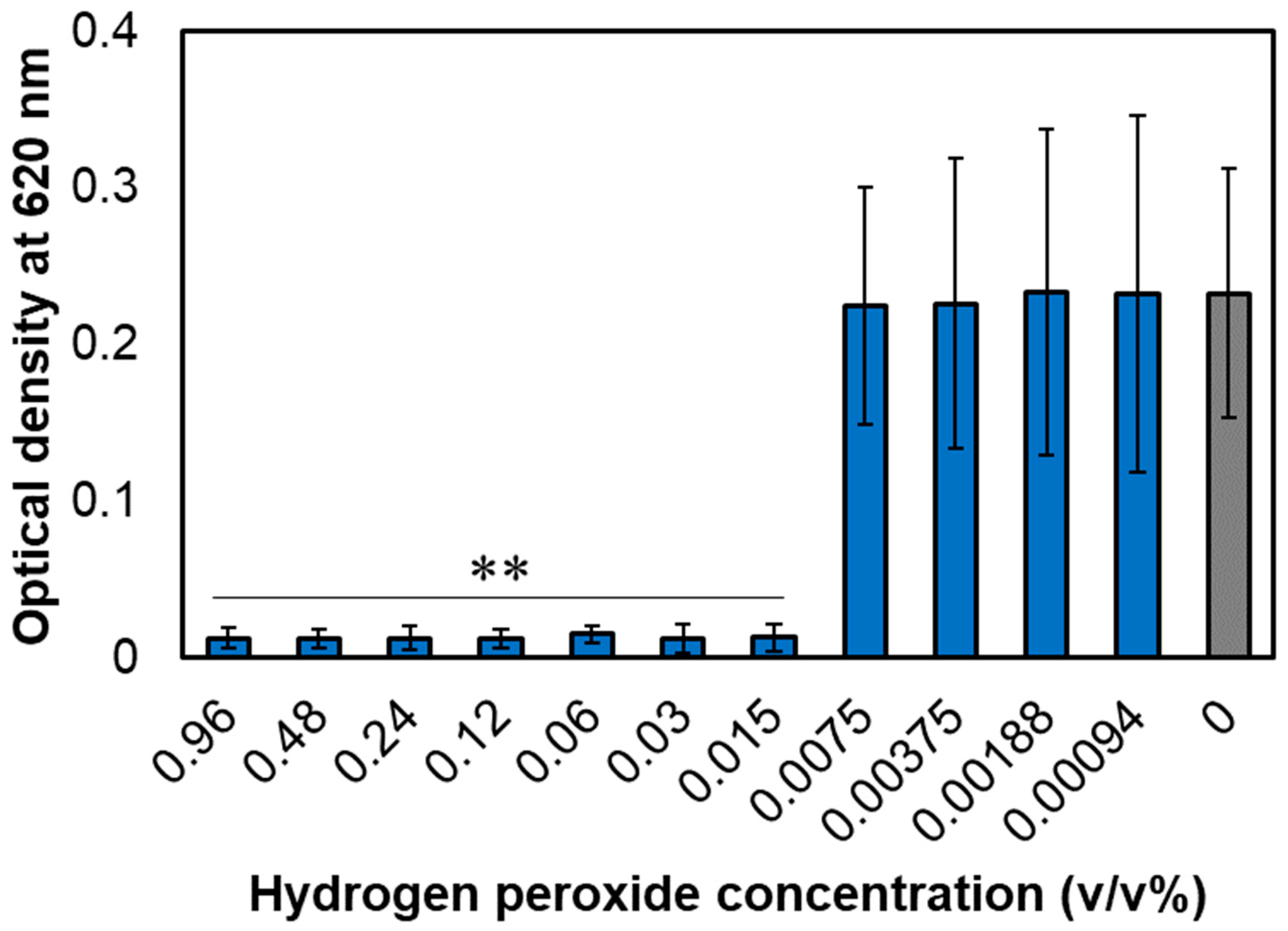
Microorganisms | Free Full-Text | Persister Cell Formation and Elevated lsrA and lsrC Gene Expression upon Hydrogen Peroxide Exposure in a Periodontal Pathogen Aggregatibacter actinomycetemcomitans

SOLVED: A solution of hydrogen peroxide is 30.0% H2O2 by mass and has a density of 1.11 g/cm³. Calculate the molarity of the solution. Show your calculation or explain your answer.
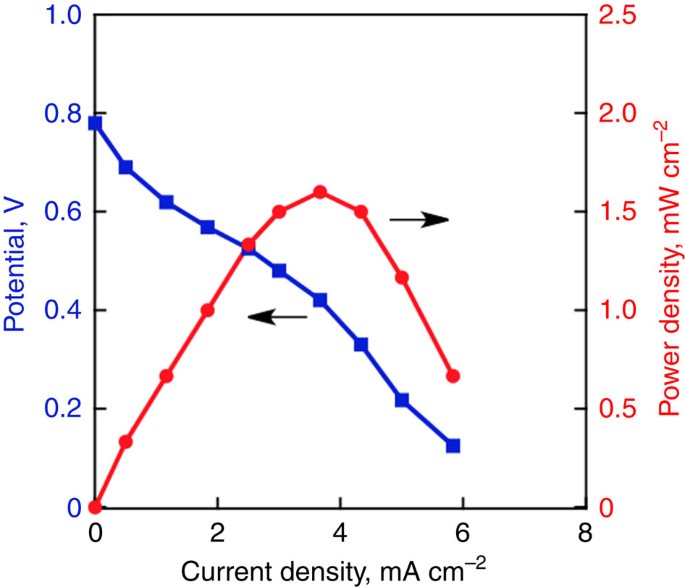
Seawater usable for production and consumption of hydrogen peroxide as a solar fuel | Nature Communications

Wink of Knowledge: Measurement of the hydrogen peroxide concentration in water with <0.025% measurement uncertainty | TrueDyne Sensors AG

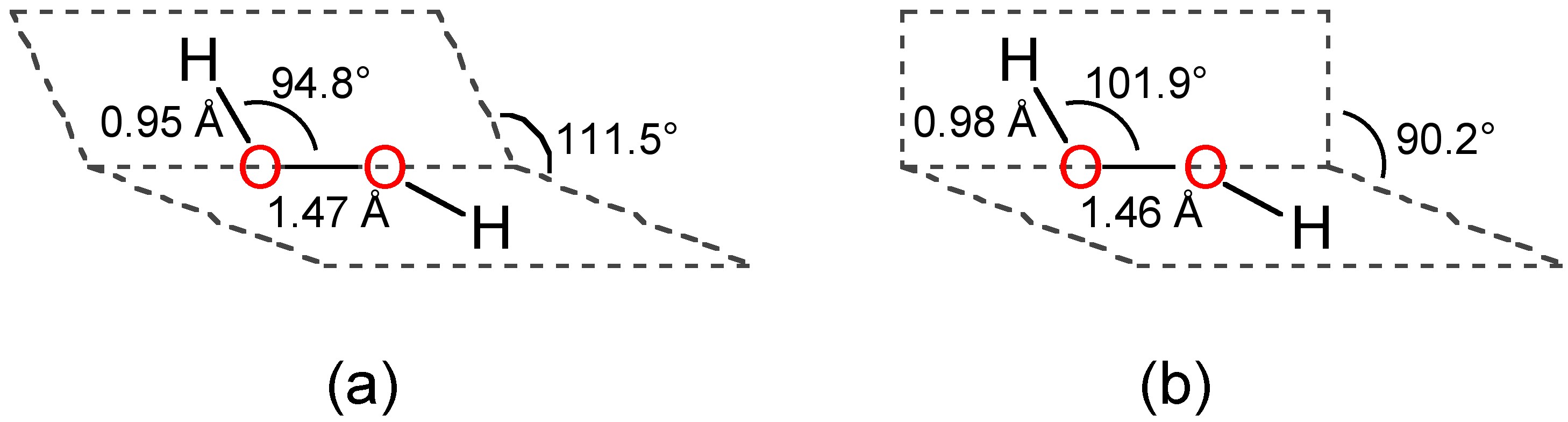
A Simple Additive Potential Model for Simulating Hydrogen Peroxide in Chemical and Biological Systems | Journal of Chemical Theory and Computation

A solution of hydrogen peroxide is 15.2% by mass. What is the molarity of the solution? Assume that the - Brainly.in